Traceability and Comfort for Users
Confidence is an absolute must. Macopharma has developed a unique system providing complete traceability of blood bag systems and individual component.
Macopharma’s solutions integrate all the expertise on disposables, equipments, softwares, processing guidelines that supports healthcare professionals to provide safer and higher quality blood components, for the benefits of donors and patients
Listing of solutionsTraceability and Comfort for Users

Collection Solutions
Separation Solutions

Leukodepletion Solutions

Platelets Solutions

Phlebotomy Solutions

Pathogen Inactivation Solutions
Cord Blood Solutions

Bio Banking

Paediatric Solutions

Transfer Solutions

Extracorporeal Photopheresis Solutions

Innov

Pathogen Inactivation Solutions

Platelets Solutions

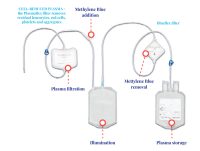
Plasma Solutions

Confidence is an absolute must. Macopharma has developed a unique system providing complete traceability of blood bag systems and individual component.
40 years of listening, understanding, and caring in every kit we produce.
Our knowledge and expertise enable us to provide with the highest quality and accompany you everyday in the blood collection process.



From automated systems that streamline the blood separation to the wide range of blood processing kits, our solutions optimize your workflow, provide consistent blood products and improve patients outcome.



A key step to deliver several benefits to patients receiving transfusions.
As a pioneer in in-line filtration, we are proud to share our experience in blood filtration.



As one of the main actors in the promotion of pool platelet concentrates from Buffy Coat, Macopharma offers you a complete solution to prepare high quality and safe blood components.
The Buffy Coat method allows optimization and adding value to the blood components obtained from wholeblood, providing great reduction of leukocytes in the final products, leading to significant reduction of adverse transfusion reactions.


For patients requiring therapeutic phlebotomy, we offer specific collection sets.
Standing at the forefront, constantly pushing boundaries and setting new industry standards, we have developed advanced pathogen inactivation technologies to provide patients with high safety blood components.


FROM BIRTH TO STEM CELLS PRESERVATION, make the best out of every drop of cord blood.
At Macopharma, we understand that each birth is a unique opportunity to save lives.
With our extensive expertise in blood donation and component processing, we provide a trusted, all-encompassing solution for cord blood banks, covering everything from cord blood collection to stem cell extraction and cryopreservation.




Macopharma offers a comprehensive Biobanking solution designed to ensure the safe and efficient preservation of human cells and tissues for therapeutic use.




For patients with specific needs, we manufacture devices specifically designed to help blood centres provide dedicated care to most vulnerable patients.
Splitting, transferring, pooling, sampling, for any step of blood processing, we offer a variety of volumes and configurations for blood centres, hospitals and research laboratories.
Over the last years, extracorporeal photopheresis (ECP) has been recognized by the scientific community as therapeutic option to treat various disorders.
Recommendations from a UK group of photopheresis experts summarize the positive impact of the technique on the treatment on several pathologies.¹
(1) Scarisbrick et al. U.K. consensus statement on the use of extracorporeal photopheresis for treatment of cutaneous T-celllymphoma and chronic graft-versus-host disease.2008.
We believe in innovations to help our company to reach the vision of raising the standards of care.
Across the organization, we organize to develop collaboratively new products, new services, new processes…



Standing at the forefront, constantly pushing boundaries and setting new industry standards, we have developed advanced pathogen inactivation technologies to provide patients with high safety blood components.






A full range of solutions that contribute to better health outcomes.
The Buffy Coat processing will enable us to make the best from every drop of blood collected.





An inactivation solution for plasma units from whole blood and Apheresis



